Left ventricular assist devices (LVADs) have moved from being a bridge to a heart transplant to destination therapy for patients with severe heart failure. Although their use in the general public has increased, they still provide a challenge to the emergency medicine (EM) physician. This series aims to cover the basics of how the EM physician approaches the care of these patients.
Left ventricular assist devices (LVADs) have moved from being a bridge to a heart transplant to destination therapy for patients with severe heart failure. Although their use in the general public has increased, they still provide a challenge to the emergency medicine (EM) physician. This series aims to cover the basics of how the EM physician approaches the care of these patients.
Some important numbers to consider:
- 5.7 million patients in the USA have heart failure, half of which will die within 5 years [1].
- LVAD use is expanding with over 22,000 being placed to date.
- With a presentation rate to the hospital of 3 per patient LVAD year [2,3].
Bleeding
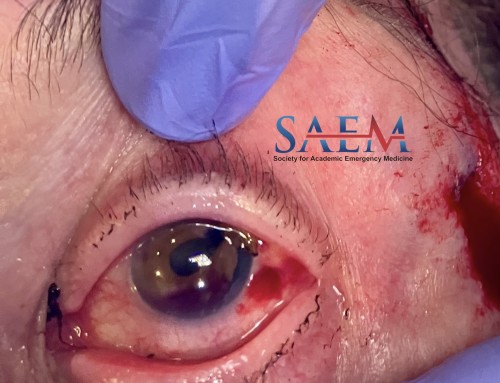
Because LVAD patients are anticoagulated, coagulopathy is a very common complication. Many patients present with gastrointestinal bleeds (GIB) because of both pharmacologic anticoagulation and an acquired Von Willebrand Factor (vWF) deficiency caused by the continuous flow LVAD shear stress [8]. Vascular malformations have also been reported to develop in LVAD patients because of mechanisms that are still not completely understood and are best diagnosed with angiography.
Reversing Anticoagulation:
- Should be done in consultation with the inpatient LVAD team
- It is associated with a significant increase in 90 thrombotic event [9].
- The “middle ground” with some sources recommending giving platelets, small amounts of fresh frozen plasma, Vitamin K, or desmopressin initially [5].
- Administration of Factor VII or prothrombin complex concentrate are a last resort, reserved for the coding LVAD patient [5].
ALWAYS talk to the LVAD team before reversing anticoagulation because reversal can cause acute pump thrombus which carries a very high mortality and requires LVAD explant and replacement. It is always acceptable to withhold further doses of home regimen anticoagulation while in the ED when presenting with GIB. Otherwise, treat LVAD patients who have a GIB just like any other GIB patients: serial H&H, type and cross, transfuse to HgB of 7 g/dL, and schedule emergent endoscopy.
Infection
Many VAD patients will have an infection associated with their device and its components. Driveline infections are estimated to occur in 17-30% of patients [5]. Ultrasound and computed tomography (CT) can help elucidate the severity.
- Typical organisms are Staphylococcus aureus, Pseudomonas species, Enterococcus species, Enterobacter species, and Candida species.
- Pocket infections (inside the pocket created for the LVAD) typically present as an abscess near the skin and require surgical drainage and debridement. Fortunately LVAD devices and cannula infections are fairly uncommon but carry high mortality rates. Suspect cannula infection in a patient who appears septic with no definitive source.
- Maintain doppler MAP above 60 mmHg; use vasopressor agents if needed. Obtain blood cultures, and investigate possible sources. Admit if systemically infected, and begin broad-spectrum antibiotics [5,10].
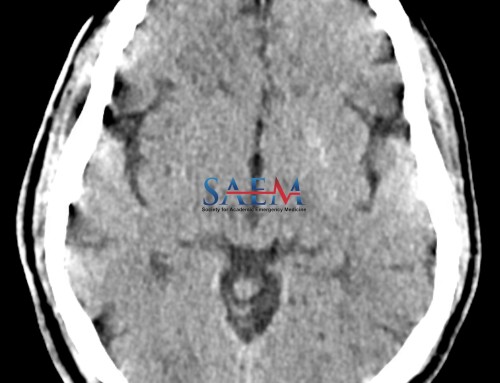
Ischemic and Hemorrhagic Stroke
LVAD patients have an increased risk of ischemic and hemorrhagic stroke compared to the general population.
- Ischemic strokes are more common than hemorrhagic strokes and carry high morbidity and mortality in LVAD patients.
- Hemorrhagic strokes, other than small, traumatic subarachnoid hemorrhages, are often catastrophic with more than half of patients dying during that hospitalization.
- Standard CT head is still the initial test of choice for stroke and can be followed by a CT perfusion scan or CT angiography for ischemic strokes.
Risk factors for ischemic stroke in the LVAD population include the standard list (history of stroke, atrial fibrillation, smoking, hyperlipidemia, diabetes, etc) as well as acquired vWF deficiency and their life long anticoagulation status. Additionally, systemic infection has been associated with a 2-fold increase in ischemic stroke risk. Interestingly, non-compliance or discontinuation of anticoagulation therapy has been inconsistently associated with increased risk of stroke [11,12].
Treatment of LVAD patients who are having a stroke:
- Do NOT give aspirin or other anticoagulants.
- Work with the inpatient team to transition them from their oral anticoagulation to unfractionated heparin.
- Thrombolytics are contraindicated.
- Endovascular catheter-directed thrombolytic therapy is a potential option for small infarcts.
It bears repeating that emergency physicians should not reverse anticoagulation without first speaking with the LVAD team. There are LVAD adjustments that can be made by the LVAD team if reversal is required, and the decision to reverse should be a multidisciplinary discussion. Blood pressure control in hemorrhagic stroke is poorly defined but some centers use a benchmark MAP of less than 90 mmHg. A team at Columbia University has developed an algorithm for the management of acute neurological deficits in LVAD patients [13].
Arrhythmia
As discussed above, an ECG is reliable for overall rhythm in LVAD patients.
- Tachyarrhythmias are common in VAD patients, and ventricular arrhythmias are of greatest concern.
- Ventricular arrhythmias are most likely to occur within the first month of implantation and are associated with a more difficult course and increased overall mortality [14].
- The urgency of intervention should be based upon the clinical presentation since the VAD can support patients temporarily through otherwise fatal arrhythmias [15].
Stable patients can be medically managed, even patients in VF/VT.
- The concern with ventricular arrhythmia is a loss of forward flow from the right heart (not supported by a VAD) which results in low LVAD flow and increased risk of suction events, leading to hypotension and hemodynamic compromise.
- Ventricular arrhythmias can worsen right heart function [4].
- Unstable ventricular arrhythmia patients should be defibrillated exactly like any other patient. Do NOT disconnect the controller from the driveline to defibrillate.
- All currently available LVADs in the US can sustain the shock.
Hypovolemia
LVAD patients are preload dependent. Dehydration is common, so diuretics and nitrates should be used cautiously since they reduce preload. The LVAD may show ‘suction event’ or ‘low flow’ alarms. A suction event is a collapse of the ventricular septum onto the LVAD inflow tract. Look for causes of low volume, commonly hemorrhage owing to the anticoagulation. Titrate volume resuscitation to doppler MAP of 70 mmHg [4].
Hypotension/Shock
Doppler MAPs less than 60 mmHg are considered hypotensive. Cardiogenic shock will present the same in LVAD patients as any other patients with signs of end organ dysfunction and poor perfusion. Inotropic agents may be used to support these patients. Options include milrinone, dobutamine, and epinephrine, although we recommend epinephrine as a first line agent for the undifferentiated LVAD shock patient. There is no evidence to support one over another, however. Vasoconstrictive agents may be used to support a low MAP as well, especially in distributive shock.
Did you miss the LVAD introduction, LVAD diagnostic evaluation or LVAD Complications? Stay tuned for upcoming and last post in the LVAD series, The Crashing LVAD Patient!
For an on-the-spot clinical reference, use ALiEM’s Paucis Verbis card for LVAD complications.
References:
- Heart Failure Factsheet. Division for Heart Disease and Stroke Prevention. Published June 16, 2016. Accessed June 11, 2018.
- Kroekel PA, George L, Eltoukhy N. How to Manage the Patient in the Emergency Department With a Left Ventricular Assist Device. Journal of Emergency Nursing. 2013;39(5):447-453. PMID 22595685
- Vierecke J, Schweiger M, Feldman D, et al. Emergency procedures for patients with a continuous flow left ventricular assist device. Emergency Medicine Journal. 2016;34(12): 831-841. PMID 27852651
- Pistono M, Corrà U, Gnemmi M, Imparato A, Temporelli PL, Tarro Genta F, et al. How to face emergencies in heart failure patients with ventricular assist device. Int J Cardiol. 2013 Oct 15;168(6):5143–8. PMID 23992932
- Sen A, Larson JS, Kashani KB, Libricz SL, Patel BM, Guru PK, et al. Mechanical circulatory assist devices: a primer for critical care and emergency physicians. Crit Care. 2016 Jun 25;20(1):153. PMID 27342573
- Yuan N, Arnaoutakis GJ, George TJ, et al. The spectrum of complications following left ventricular assist device placement. J Card Surg 2012;27:630-8. PMID 22978843
- Kilic A, Acker MA, Atluri P. Dealing with surgical left ventricular assist device complications. J Thorac Dis. 2015 Dec;7(12):2158–64. PMID 26793336
- Goldstein DJ, John R, Salerno C, et al. Algorithm for the diagnosis and management of suspected pump thrombus. J Heart Lung Transplant 2013;32:667–70. PMID 23796150
- Suarez J, Patel CB, Felker GM, et al. Mechanisms of bleeding and approach to patients with axial-flow left ventricular assist devices. Circ Heart Fail 2011;4:779–84. PMID 22086831
- Wasson LT, Yuzefpolskaya M, Wakabayashi M, et al. Hypertension: an unstudied potential risk factor for adverse outcomes during continuous flow ventricular assist device support. Heart Fail Rev 2015;20:317–22. PMID 25283767
- Willey JZ, Demmer RT, Takayama H, Colombo PC, Lazar RM. Cerebrovascular disease in the era of left ventricular assist devices with continuous flow: risk factors, diagnosis, and treatment. J Heart Lung Transplant. 2014 Sep;33(9):878–87. PMID 24997495
- Stulak JM, Lee D, Haft JW, et al. Gastrointestinal bleeding and subsequent risk of thromboembolic events during support with a left ventricular assist device. J Heart Lung Transplant 2014;33:60-4. PMID 24021944
- Lampert BC, Eckert C, Weaver S, et al. Blood pressure control in continuous flow left ventricular assist devices: efficacy and impact on adverse events. Ann Thorac Surg 2014;97:139-46. PMID 24075484
- Bedi M, Kormos R, Winowich S, McNamara DM, Mathier MA, Murali S. Ventricular arrhythmias during left ventricular assist device support. Am J Cardiol 2007;99: 1151–3. PMID 17437746
- Andersen M, Videbaek R, Boesgaard S, Sander K, Hansen PB, Gustafsson F. Incidence of ventricular arrhythmias in patients on long-term support with a continuous-flow assist device (HeartMate II). J Heart Lung Transplant 2009;28:733–5. PMID 19560703